SAR Analysis in Research: A Complete Guide to Structure Activity Relationships
- Home
- Research Services
- Systematic Review
- SAR Analysis in Research: A Complete Guide to Structure Activity Relationships
High-Impact Journals
- Understanding Structure Activity Relationships (SAR)
- Key Components of SAR Analysis
- SAR Workflow in Drug Discovery
- Types of SAR Approaches
- Importance of SAR in Research
- Applications of SAR
- Challenges in SAR Analysis
- Best Practices for Effective SAR Studies
- Example of SAR Table
- Future Trends in SAR Research
- Conclusion
Interesting topics
SAR Analysis in Research: A Complete Guide to Structure Activity Relationships
The Structure-Activity Relationship (SAR) study is an essential part of contemporary pharmaceutical research and medicinal chemistry, widely recognised as a core component of SAR analysis in research. The SAR investigation aims to determine how the chemical structure affects the biological activity of the compound. Thus, by altering the chemical structure, it is possible to obtain substances with enhanced biological activity that could be used in medicinal practice, particularly in SAR in pharmaceutical research.[1]
1. Understanding SAR in Research
SAR is the correlation between the structure of the compound and its pharmacological activity. It entails the examination of the effect of changing functional groups, the stereochemistry, and the size of the molecules on the binding ability, activity, selectivity, and toxicity of the molecule. SAR is commonly applied in drug design and is closely linked with statistical analysis in drug discovery.
The main idea in SAR is that even the slightest change in the chemical structure of the compound can result in drastic changes in biological activity. For instance, the change from a hydrogen atom to a methyl group can improve the lipophilicity of the compound, hence enhancing drug effectiveness through increased permeability. However, this change can also make the drug more toxic, highlighting the importance of medical data analysis in research.
2. Key Components of SAR Analysis
Component | Description |
Functional Groups | Specific groups of atoms that influence reactivity and binding |
Molecular Size | Affects the ability to fit into biological targets |
Shape and Geometry | Determines interaction with receptors |
Electronic Properties | Influence bonding and molecular interactions |
Lipophilicity | Impacts absorption and distribution |
These components collectively determine how a molecule interacts with its biological target, such as an enzyme or receptor.
3. SAR Workflow in Drug Discovery
The SAR approach usually starts with identifying the lead molecule. The lead molecule is the initial compound from which various modifications are made to optimise its structure. Analogues of the lead molecule are synthesised and tested for biological activity using experimental and clinical data statistical analysis approaches.[2]
Patterns can be established based on the analysis of data obtained from experiments to establish the correlation between structures and activities. Such information can then be used to further modify the structure of the molecule until the desired properties are achieved.
4. Types of SAR Approaches
SAR methods may be classified as qualitative or quantitative. In qualitative SAR studies, structural modifications are observed to understand their effect on activity. In quantitative SAR (QSAR), mathematical models are used to predict biological activity, forming an important part of statistical analysis in drug discovery.[3]
QSAR models incorporate parameters such as hydrophobicity, electronic effects, and steric factors to build predictive relationships. These models are essential in modern clinical research statistics, and computational drug design.

5. Importance of SAR in Research
SAR analysis plays a critical role in multiple stages of drug discovery. It helps in selecting optimal compounds, enhancing efficacy, and reducing side effects. Through SAR insights, researchers can design drugs with improved specificity and reduced off-target effects, which is a key objective in SAR in pharmaceutical research.
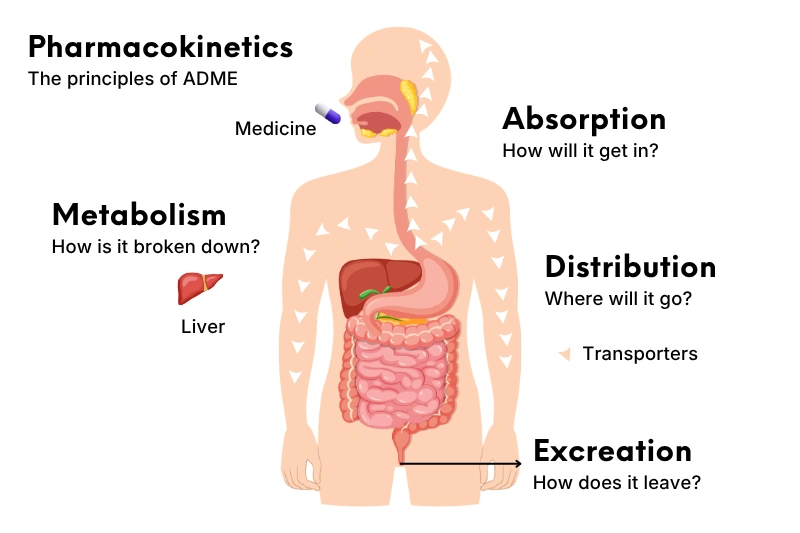
Additionally, SAR contributes to improving ADME (Absorption, Distribution, Metabolism, Excretion) properties. These improvements ensure that drugs effectively reach their biological targets, supported by medical data analysis in research.
6. Applications of SAR
SAR finds applications in diverse fields, including pharmaceuticals and biomedical research. In drug development, it is used for optimising drugs and for developing novel therapeutic agents. It is also used in the study of the toxicity of substances through predicting their safety profiles. Moreover, it finds application in environmental studies in studying the effects of pollution. [4]
Moreover, SAR is crucial in studying ligand-receptor interactions, enzyme inhibition, and mechanisms of drug action. These applications are supported by clinical research statistics methods, and data-driven modelling approaches.
7. Challenges in SAR Analysis
SAR analysis faces some obstacles despite having various benefits. First, biological systems are complex since there are many determinants that contribute to activity. Second, the connection between activity and structure does not necessarily have to be linear and therefore cannot be predicted.[5]
Another major challenge is the requirement for large, high-quality datasets. Data collection is time-consuming and resource-intensive, and experimental variability can impact consistency. These challenges highlight the importance of robust clinical data statistical analysis.
8. Best Practices for Effective SAR Studies
For any research work to bear fruit, there should be a systematic procedure for carrying out a SAR study. This involves choosing suitable lead molecules, creating well-designed analogues, and conducting credible experiments. [6]
The integration of computational tools with experimental methods significantly enhances SAR efficiency. Machine learning and modelling techniques enable advanced statistical analysis in drug discovery, improving prediction accuracy and decision-making.
8.1. Example of SAR Table
| Compound | Structural Modification | Activity Level | Observations |
| A | Base structure | Moderate | Initial lead compound |
| B | Added methyl group | High | Increased potency |
| C | Hydroxyl substitution | Low | Reduced activity |
| D | Halogen substitution | Very High | Improved binding affinity |
This table illustrates how structural changes influence biological activity, a core principle of SAR analysis.
9. Future Trends in SAR Research
Future developments in SAR include the integration of artificial intelligence and big data analytics. AI models can process vast datasets to predict drug behaviour, transforming SAR in pharmaceutical research. [7]
Advances in structural biology and high-throughput screening are also enhancing SAR efficiency. These technologies, combined with clinical research statistics, are accelerating drug discovery processes.
Connect with us to explore how we can support you in maintaining academic integrity and enhancing the visibility of your research across the world!
Conclusion
Structure-Activity Relationship studies are fundamental to modern pharmacological research. SAR enables scientists to design safer and more effective drugs through systematic analysis of structure and activity relationships. Despite challenges, advancements in computational tools and clinical research statistics methods continue to expand the potential of SAR analysis.
SAR Analysis in Research: A Complete Guide to Structure Activity Relationships. Our Pubrica consultants are here to guide you. [Get Expert Publishing Support] or [Schedule a Free Consultation]
References
- Chan, L.Y.H., Morris, S.E., Stockwell, M.S. et al.Estimating the generation time for SARS-CoV-2 transmission using United States household data, December 2021–May 2023. Sci Rep (2026). https://doi.org/10.1038/s41598-026-46596-6
- Hussein, M. A., & Munirathinam, G. (2026). Artificial Intelligence-Driven Natural Product Discovery for Cancer Metastasis and Chemoresistance: From Computational Prediction to Preclinical Validation. Cancers, 18(5), 719. https://doi.org/10.3390/cancers1
- Acosta-Murillo, R., Ortiz-Bayliss, J. C., & Zapata-Morin, P. A. (2026). Enhancing cancer drug discovery: QSAR modeling with machine learning and chemical representations. PloS one, 21(3), e0343654. https://doi.org/10.1371/journal.pone
- Bononi, G., & Granchi, C. (2026). Recent Advances in Development of Small Molecules to Fight Cancer-Second Edition. Molecules (Basel, Switzerland), 31(5), 808. https://doi.org/10.3390/molecules
- Landi, A., Mimouni, Y., Demoisy, A. et al.Ethics and regulatory guidance to address the challenges encountered in research on rare diseases in a multinational dimension: the experience of a European funded programme. BMC Med Ethics (2026). https://doi.org/10.1186/s12910-025-01370-7
- Herdiana, Y., Mohammed, A. F. A., Kautsar, A. P., Sopyan, I., & Wathoni, N. (2026). Artificial Intelligence as a Disruptive Force in Pharmaceutical Innovation: Transforming Discovery, Development, and Manufacturing. Drug design, development and therapy, 20, 588295. https://doi.org/10.2147/DDDT.S588
- Chen, R., Tang, Y., Duan, L., Jiang, Z., & Zhu, M. (2026). Global Trends and Hotspot Analysis of Nanomaterials in the Field of Acne: A Bibliometric Perspective. Clinical, cosmetic and investigational dermatology, 19, 586477. https://doi.org/10.2147/CCID.S586







