- Services
- Discovery & Intelligence Services
- Publication Support Services
- Sample Work

Publication Support Service
- Editing & Translation
-
Editing and Translation Services
- Sample Work

Editing and Translation Service
-
- Research Services
- Sample Work

Research Services
- Physician Writing
- Sample Work

Physician Writing Service
- Statistical Analyses
- Sample Work

Statistical Analyses
- Data Collection
- AI and ML Services
- Medical Writing
- Sample Work

Medical Writing
- Research Impact
- Sample Work

Research Impact
- Medical & Scientific Communication
- Medico Legal Services
- Educational Content
- Academic Editorial Services
- Educational Editorial Service
-
Education Editorial Services
-
- Industries
- Subjects
- About Us
- Academy
- Insights
- Contact Us
The Role of Biostatistics in Clinical Research Programs
- Home
- Academy
- Poster Presentation
- The Role of Biostatistics in Clinical Research Programs
Research Impact
- Importance of Biostatistics in Clinical Research
- Role of Biostatistics Across Clinical Trial Phases
- Study Design and Sample Size Determination
- Data Collection and Statistical Analysis
- Regulatory Compliance and Reporting
- Challenges in Biostatistics for Clinical Research
- Skills Required for Biostatisticians
- Future of Biostatistics in Clinical Research
- Conclusion
Interesting topics
The Role of Biostatistics in Clinical Research Programs
1. Importance of Biostatistics in Clinical Research
2. Role of Biostatistics Across Clinical Trial Phases
3. Study Design and Sample Size Determination
4. Data Collection and Statistical Analysis
5. Regulatory Compliance and Reporting
6. Challenges in Biostatistics for Clinical Research
7. Skills Required for Biostatisticians
8. Future of Biostatistics in Clinical Research
9. Conclusion
Biostatistics are critical to the success of most clinical research projects because they help ensure the collection, analysis and accurate interpretation of scientific information. The application of statistical techniques and methodologies to the design of clinical trials and evaluation of treatments provides the mathematical and analytical basis for evidence-based medicine. Without appropriate statistical planning and analysis, the results of clinical research will be utilised incorrectly, which will result in erroneous conclusions and an increase in the risk to patient safety in biostatistics in clinical research.[1]
In addition to being an essential element of the clinical research process, biostatisticians work closely with clinical researchers, regulatory agencies and other healthcare professionals to ensure that the research studies meet scientific and regulatory standards. Their efforts help to convert raw clinical data into useful and meaningful insights through biostatistical data analysis in research, which can be used to inform decision-making in drug development and health care policy, often supported by biostatistics consulting services and statistical programming services.[2]
1. Importance of Biostatistics in Clinical Research
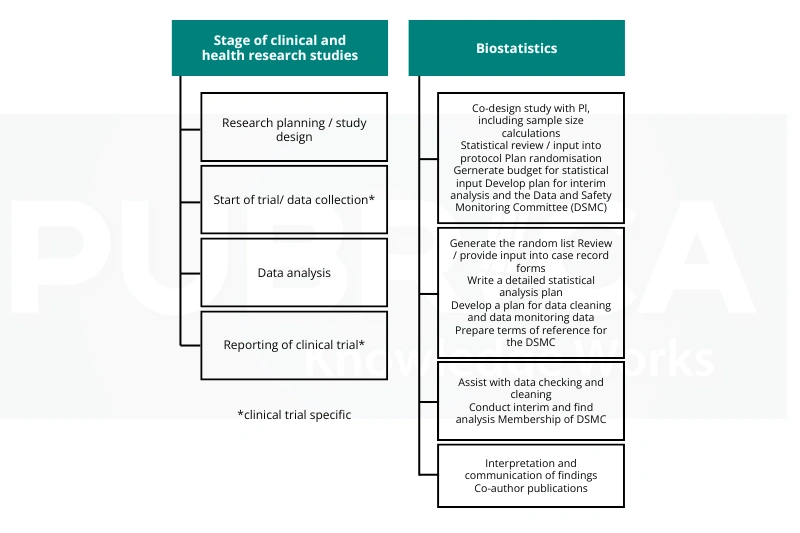
Biostatistics plays an essential role in clinical research; it is used extensively to validate, verify and interpret data collected from clinical trials (clinical research). Biostatistics is used for clinical trial design by researchers; therefore, biostatistics in clinical research provides the basis for making the decision about whether there is enough evidence to support the safety and effectiveness of evidence obtained from clinical trial outcomes through proper statistical planning in clinical research.[3]
The Importance of Biostatistics:
- Provides accurate and reliable data from clinical trials using biostatistical data analysis in research.
- Provides evidence-based decision-making.
- Assists with the determination of sample size and statistical power.
- Evaluates whether the outcome and effectiveness of treatments are safe and effective.
- Guides regulatory approvals and submissions.
Biostatistics lends validity and credibility to research and gives health care providers the ability to make informed treatment decisions based on the evidence they have gathered from their patients.
2. Role of Biostatistics Across Clinical Trial Phases
A biostatistician plays a role in all stages of clinical research, including designing the protocol, carrying out data analysis, and preparing for regulatory submission in the final report across different clinical trial phases. This is done in conjunction with the research team by assisting them in the design of protocols, performing data analyses through statistical programming services, and providing regulatory submission-ready tables of results.[4]
Biostatistics Contributions in Each Phase
Clinical Trial Phase | Role of Biostatistics | Outcome |
Phase I | Safety and dosage analysis | Determines safe dosage levels |
Phase II | Efficacy assessment | Evaluates treatment effectiveness |
Phase III | Large-scale statistical validation | Confirms safety and efficacy |
Phase IV | Post-marketing surveillance | Monitors long-term effects |
These statistical analyses help researchers draw reliable conclusions and ensure that treatments meet regulatory standards before reaching the market.
3. Study Design and Sample Size Determination
Biostatistics plays one of the most important roles in clinical research when it comes to designing studies, which includes determining sample size. If a study was poorly constructed, this could lead to biased results and/or insufficient evidence to support the conclusions of the study.[5]
Biostatisticians consider the following when designing the clinical study:
- Research Objectives and Hypotheses
- Type of data (i.e., qualitative vs. quantitative)
- Characteristics of the study participants
- Level of statistical significance required
- Expected variability in results
The purpose of calculating an accurate sample size is to ensure that clinical studies have enough subjects so that statistically significant differences can be found between the two treatment groups without using unnecessary resources.
4. Data Collection and Statistical Analysis
Data Collection, Data Management Planning & Data Analysis through Statistical Methods Utilising Biostatistics ensures strong biostatistical data analysis in research. Biostatistics guarantees that data collection is standardised correctly for analysis. Biostatisticians will implement the development of the data management plan, selection of appropriate statistical tests, and interpretation of results using advanced statistical programming services.[6]
Some Statistics Methods Commonly Used are:
- Descriptive statistics for describing populations
- Inferential statistics for testing hypotheses/population differences
- Using regression analysis to determine the relationship between variables
- Utilising survival analysis to evaluate time-to-event outcomes
- Performing meta-analyses to summarise research findings
This will allow for valid conclusions drawn from studies conducted using credible methods and scientifically validate them.
5. Regulatory Compliance and Reporting
Biostatistics plays an important role in preparing regulatory submissions for health authorities, including the FDA and EMA. Statistically reliable evidence generated through biostatistics in clinical research is necessary for regulatory agencies to grant approval for new drugs, medical devices, and treatment protocols.[7]
Biostatisticians ensure that the following occurs:
- Correctly follows statistical regulations when analysing data
- Clearly presents results in clinical study reports
- Documents statistical methods appropriately
- Provides reproducible and transparent results
Accurate statistical reporting enhances a company’s ability to receive regulatory approvals and comply with international standards of research.
6. Challenges in Biostatistics for Clinical Research
Biostatistics plays an essential role in supporting clinical research but has several obstacles to overcome to do so effectively even with modern biostatistics services. These obstacles include the inability to manage large, complex datasets; inability to assure data quality and consistency; inability to deal with missing or incomplete datasets; inability to adjust to changing statistical methodologies; and inability to meet tight deadlines for clinical research.[8]
7. Skills Required for Biostatisticians
Biostatisticians who excel have both technical expertise and analytical ability in conjunction with an understanding of statistical theory & clinical trial methods and experience in biostatistics in clinical research:[9]
- Advanced knowledge of statistics
- SAS, R, SPSS skill
- Ability to interpret and visualise data
- Knowledge of clinical trial design
- Ability to communicate and cooperatively work with other disciplines.
8. Future of Biostatistics in Clinical Research
The field of biostatistics is changing as the technology and science of data continue to advance. With artificial intelligence (AI), machine learning (ML), and the increasing use of real-world data, there are greater opportunities than ever before to improve the results of clinical trials through advanced biostatistical data analysis in research. AI and ML allow for quick access to processed data, as well as opportunities for developing predictive models and more tailor-made treatments supported by modern biostatistics services.[10]
Biostatistical methods will remain critical in ensuring that the results of clinical trials are accurate, ethical, and scientifically valid.
Connect with us to explore how we can support you in maintaining academic integrity and enhancing the visibility of your research across the world!
Conclusion
Biostatistics is essential for the success of clinical research because of its ability to ensure that clinical trials are conducted ethically, scientifically, and statistically valid through strong biostatistics in clinical research. Biostatistics provides support to study designs (clinical trial design), data analysis (including data oversight), regulatory submissions, and evidence-based healthcare (use of data from clinical trial, etc.) through professional biostatistics consulting services and statistical programming services. Strong statistical planning in clinical research and analysis will strengthen the credibility of research, improve patient safety and support the development of effective treatments. With the changing nature of clinical research (including the importance of biostatistical components), biostatistics will become even more important to the future of clinical research, drug development and evidence-based healthcare through expanding biostatistics services.
The Role of Biostatistics in Clinical Research Programs. Our Pubrica consultants are here to guide you. [Get Expert Publishing Support] or [Schedule a Free Consultation]
Frequently asked questions
1.Why is biostatistics important in clinical research?
Biostatistics is essential in clinical research because it ensures accurate collection, analysis, and interpretation of data from clinical trials. It helps researchers evaluate the safety and effectiveness of treatments, supports evidence-based medical decisions, and ensures that study results are scientifically valid and reliable.
2. What role does a biostatistician play in clinical trials?
A biostatistician is involved in every stage of a clinical trial, from study design and sample size calculation to data analysis and regulatory reporting. They work closely with researchers and healthcare professionals to ensure proper statistical planning, accurate data interpretation, and compliance with regulatory standards.
3. How does biostatistics help in drug development?
Biostatistics provides the analytical foundation for drug development by evaluating clinical trial data, determining treatment effectiveness, and identifying potential risks. It helps regulatory agencies assess whether a drug is safe and effective, supporting approval and evidence-based healthcare decisions.
4. What statistical methods are commonly used in clinical research?
Common statistical methods include descriptive statistics, inferential statistics, regression analysis, survival analysis, and meta-analysis. These methods help researchers understand data patterns, test hypotheses, and draw meaningful conclusions from clinical research studies.
5. How do biostatistics consult services support researchers?
Biostatistics consulting services assist researchers with study design, statistical analysis, data interpretation, and regulatory reporting. Expert guidance ensures accurate results, improves research quality, and helps researchers meet publication and regulatory requirements efficiently.
References
- Tsiamalou, P., & Brotis, A. (2020). Biostatistics as a Tool for Medical Research: What are we Doing Wrong?. Mediterranean journal of rheumatology, 30(4), 196–200. https://doi.org/10.31138/mjr.30.4.196
- Bridges, B. (2024, August 5). The role of biostatistics in clinical research programs. com. https://www.warmanobrien.com/
- Barkan H. (2015). Statistics in clinical research: Important considerations. Annals of cardiac anaesthesia, 18(1), 74–82. https://doi.org/10.4103/0971-9784.148325
- Pugh, S. L., & Torres-Saavedra, P. A. (2021). Fundamental Statistical Concepts in Clinical Trials and Diagnostic Testing. Journal of nuclear medicine : official publication, Society of Nuclear Medicine, 62(6), 757–764. https://doi.org/10.2967/jnumed.120.24
- Bhardwaj, R., Agrawal, U., Vashist, P., & Manna, S. (2024). Determination of sample size for various study designs in medical research: A practical primer. Journal of family medicine and primary care, 13(7), 2555–2561. https://doi.org/10.4103/jfmpc.jfmpc
- Mishra, P., Pandey, C. M., Singh, U., Keshri, A., & Sabaretnam, M. (2019). Selection of appropriate statistical methods for data analysis. Annals of cardiac anaesthesia, 22(3), 297–301. https://doi.org/10.4103/aca.ACA
- Dunbar, P., Keyes, L. M., & Browne, J. P. (2023). Determinants of regulatory compliance in health and social care services: A systematic review using the Consolidated Framework for Implementation Research. PloS one, 18(4), e0278007. https://doi.org/10.1371/journal.pone
- Evans S. R. (2010). Common statistical concerns in clinical trials. Journal of experimental stroke & translational medicine, 3(1), 1–7. https://doi.org/10.6030/1939-067x-3.1.1
- Slade, E., Brearley, A. M., Coles, A., Hayat, M. J., Kulkarni, P. M., Nowacki, A. S., Oster, R. A., Posner, M. A., Samsa, G., Spratt, H., Troy, J., & Pomann, G. M. (2023). Essential team science skills for biostatisticians on collaborative research teams. Journal of clinical and translational science, 7(1), e243. https://doi.org/10.1017/cts.2023.676
- Lele C. (2017). Development of statistics as a discipline for clinical research: Past, present and future. Perspectives in clinical research, 8(1), 41–44. https://doi.org/10.4103/2229-3485.198548

