- Services
- Discovery & Intelligence Services
- Publication Support Services
- Sample Work

Publication Support Service
- Editing & Translation
-
Editing and Translation Services
- Sample Work

Editing and Translation Service
-
- Research Services
- Sample Work

Research Services
- Physician Writing
- Sample Work

Physician Writing Service
- Statistical Analyses
- Sample Work

Statistical Analyses
- Data Collection
- AI and ML Services
- Medical Writing
- Sample Work

Medical Writing
- Research Impact
- Sample Work

Research Impact
- Medical & Scientific Communication
- Medico Legal Services
- Educational Content
- Academic Editorial Services
- Educational Editorial Service
-
Education Editorial Services
-
- Industries
- Subjects
- About Us
- Academy
- Insights
- Contact Us
Types of pre-experimental research design?
- Home
- Academy
- Poster Presentation
- Types of pre-experimental research design?
Research Impact
- Definition of Pre-Experimental Research Design
- Key Characteristics of Pre-Experimental Research Design
- Types of Pre-Experimental Research Design
- Steps in Conducting Pre-Experimental Research
- Data Analysis in Pre-Experimental Research
- Advantages and Disadvantages of Pre-Experimental Research Design
- Limitations of Pre-Experimental Research Design
- Applications of Pre-Experimental Research Desig
- Example of a Pre-Experimental Research Study
Interesting topics
Types of pre-experimental research design?
1. Definition of Pre-Experimental Research Design
2. Key Characteristics of Pre-Experimental Research Design
3. Types of Pre-Experimental Research Design
4. Steps in Conducting Pre-Experimental Research
5. Data Analysis in Pre-Experimental Research
6. Advantages and Disadvantages of Pre-Experimental Research Design
7. Limitations of Pre-Experimental Research Design
8. Applications of Pre-Experimental Research Design
9. Example of a Pre-Experimental Research Study
The pre-experimental designs are the most fundamental of the experimental research design approaches within the broader field of research methodology. A pre-experimental design gathers evidence that supports or rejects a hypothesis related to cause-and-effect relationships, without random assignment, control groups, or full manipulation of variables.[1]
A pre-experimental research design is very limited in controlling extraneous variables, unlike quasi-experimental design and true types of experimental research design, which can properly account for such variables. For this reason, pre-experimental designs are generally used for basic research, pilot studies, procedural studies, and exploratory research. The application of pre-experimental research design is very common in educational settings and in health sciences, psychology, and social sciences, where advanced experimental research design examples may not be practical or ethical. Such designs are often recommended by research methodology help and academic research consulting services for feasibility testing. [2]
1. Definition of Pre-Experimental Research Design
Pre-experimental research designs use quantitative approaches in which the researcher studies the effects of one or more independent variables on dependent variables without randomization or control groups. This pre-experimental research design approach is widely discussed in research methodology services and is often applied in dissertation research design during early-stage investigations. [1]
2. Key Characteristics of Pre-Experimental Research Design
These characteristics distinguish pre-experimental research design from other types of experimental research design and highlight its role within overall research methodology frameworks. [3]
| Feature | Description | Example |
|---|---|---|
| Level of Control | Minimal or no control over extraneous variables | Teaching a new method to one class |
| Randomization | No random assignment of participants | Convenience sampling |
| Control Group | Often absent | Single-group studies |
| Research Nature | Quantitative and exploratory | Pilot educational intervention |
| Causal Inference | Limited ability to establish causation | Observed improvement after treatment |
3. Types of Pre-Experimental Research Design
There are three major kinds of research designs under pre-experimental research design. Researchers often use these designs to establish a foundation for later quasi-experimental design or true experimental research design studies. [4]
3.1. One-Shot Case Study Design
In the one-shot case study design, a single group is exposed to a treatment, and the outcome is measured only once after the intervention.[5]
X → O
Explanation:
- X = Treatment / Intervention
- O = Observation / Measurement (post-test only)
In this design, a single group is exposed to an intervention (X), and the outcome (O) is measured only once after the treatment, without any pretest or control group.
Limitation:
The absence of baseline data or a comparison group makes it difficult to attribute observed outcomes solely to the treatment.
3.2. One-Group Pretest–Post-test Design
This design involves measuring the dependent variable both before and after the treatment is applied to the same group.[6]
O₁ → X → O₂
Explanation:
- O₁ = Pretest observation (before intervention)
- X = Treatment / Intervention
- O₂ = Post-test observation (after intervention)
This design measures the dependent variable before and after the treatment is applied to the same group, allowing comparison of changes over time.
Strength:
Allows comparison of changes over time within the same group.
Limitation:
Observed changes may result from external factors such as maturation, history, or testing effects rather than the intervention itself.
3.3. Static Group Comparison Design
The static group comparison design compares two existing groups, one receiving the treatment and the other not, without random assignment. [7]
X → O
— → O
Explanation:
- X = Treatment / Intervention (applied to the experimental group)
- O = Observation / Measurement
- — = No treatment (control/comparison group)
In this design, two existing groups are compared one group receives the treatment and the other does not without random assignment.
Limitation:
Pre-existing differences between groups may threaten internal validity.
4. Steps in Conducting Pre-Experimental Research
The steps involved in pre-experimental research reflect standard research methodology services, guiding researchers from problem identification to data interpretation when full experimental control is not feasible. [8]
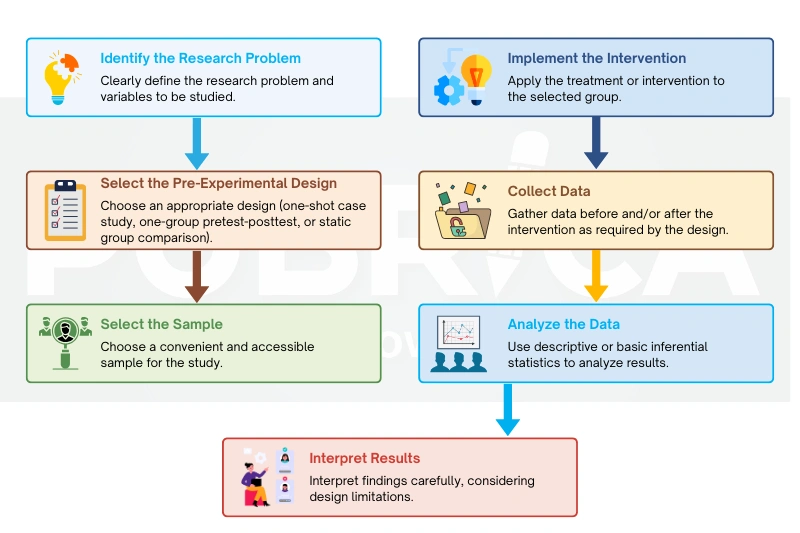
5. Data Analysis in Pre-Experimental Research
When analysing data obtained through pre-experimental studies, researchers typically use:
- Descriptive Statistics (Mean, Percent, Standard Deviation)
- Paired t-tests (often used in pretest-post-test designs)
- Graphical and comparative data analysis of outcomes
Findings are suggestive only because of the limited amount of control the researcher has over the experiment. [9]
6. Advantages and Disadvantages of Pre-Experimental Research Design
This comparison highlights why pre-experimental research design is suitable for exploratory studies but limited when compared with rigorous experimental research design and quasi-experimental design approaches.
Aspect | Advantages | Disadvantages |
Research Simplicity | Easy to design and implement | Lacks methodological rigor |
Cost & Time | Cost-effective and time-efficient | Limited depth of analysis |
Control of Variables | Useful when control is not possible | No control over extraneous variables |
Randomization | Suitable without random assignment | No randomization, causing selection bias |
Control Group | Useful when control groups are impractical | Control group usually absent |
Causal Inference | Helps explore possible relationships | Cannot establish true cause-and-effect |
Validity | Helpful for preliminary validation | High threat to internal validity |
Research Purpose | Ideal for pilot and exploratory studies | Not suitable for confirmatory research |
Ethical Feasibility | Ethical in sensitive research contexts | Ethical concerns may still exist |
Generalizability | Useful for initial insights | Limited generalizability |
7. Limitations of Pre-Experimental Research Design
The above diagram describes the sequence of significant deficiencies of pre-experimental research design, with respect to how absence of random assignment, and non-existence of control groups leads to selection bias, limited causal relations, and potential threats to internal validity. Additionally, the limitations of pre-experimental designs limit the ability for generalisability and confirmatory power for these studies.[10]
8. Applications of Pre-Experimental Research Design
Pre-experimental research design is widely applied across disciplines and is frequently supported by research methodology services when testing new interventions before conducting large-scale experimental studies: [1]
- Education: To evaluate new methods of teaching.
- Healthcare: To conduct pilot tests of health interventions.
- Psychology: To conduct initial studies on behavioural interventions.
- Business: To evaluate training and development programs.
- Social Science: To conduct preliminary studies using a community-based approach.
These types of studies can assist researchers in deciding if an experimental design would be appropriate for their research project.
9. Example of a Pre-Experimental Research Study
A pre-experimental research study uses a one-group pretest–post-test design to obtain preliminary evidence about an intervention. Such experimental research design examples are common in healthcare and education when randomization is not possible.
Study Title
Effect of a New Antihypertensive Drug on Blood Pressure
Design Used
True experimental design (Randomized Controlled Trial – RCT)
Outcome
Patients receiving the new drug showed a significant reduction in blood pressure compared to the control group.
Connect with us to explore how we can support you in maintaining academic integrity and enhancing the visibility of your research across the world!
Conclusion
pre-experimental research design services form the basis of the research method by allowing researchers the ability to identify possible cause-effect relationships with a minimum of resources and control. Although pre-experimental designs do not provide enough rigor to reach or substantiate definite causal conclusions, they are invaluable for pilot studies, to evaluate feasibility and to develop hypotheses. Through careful application and cautious interpretation, pre-experimental designs are an important component of many research studies that will lead to more adequate experimental and quasi-experimental designs.
Strengthen your research with expert guidance. Get professional support in research methodology and experimental design from experienced academic consultants. [Get Expert Publishing Support] [Schedule a Free Consultation]
Frequently asking questions
1. What are the types of pre-experimental research design?
One-Shot Case Study (treatment then post-test), the One-Group Pretest-Posttest Design (pretest, treatment, post-test), and the Static-Group Comparison (treatment group vs. comparison group, both post-tested, but without random assignment)
2. What is one-group pretest-posttest design and its limitations?
measures a single group’s outcome (dependent variable) before (pretest) and after (posttest) an intervention to assess change, like a school’s reading scores before and after a new program.
3. How does pre-experimental research design differ from quasi-experimental design?
Pre-experimental designs are the weakest, often lacking control groups or pretests (e.g., one-shot case study), offering minimal causal evidence, while quasi-experimental designs are stronger by including comparison groups but still missing random assignment.
4. What are the main internal validity threats in pre-experimental research design?
History (external events), Maturation (participant changes over time), Testing (pretest influence), Instrumentation (measurement changes), and Statistical Regression (extreme scores shifting).
5. What is static-group comparison design in pre-experimental research?
compares an existing group that receives a treatment (experimental group) with one that doesn’t (control group), measuring outcomes after the intervention, but without random assignment.
References
- Miller, C. J., Smith, S. N., & Pugatch, M. (2020). Experimental and quasi-experimental designs in implementation research. Psychiatry research, 283, 112452. https://doi.org/10.1016/
- Nianogo, R. A., Benmarhnia, T., & O’Neill, S. (2023). A comparison of quasi-experimental methods with data before and after an intervention: an introduction for epidemiologists and a simulation study. International journal of epidemiology, 52(5), 1522–1533. https://doi.org/10.1093/ije/dyad032
- Harris, A. D., McGregor, J. C., Perencevich, E. N., Furuno, J. P., Zhu, J., Peterson, D. E., & Finkelstein, J. (2006). The use and interpretation of quasi-experimental studies in medical informatics. Journal of the American Medical Informatics Association : JAMIA, 13(1), 16–23. https://doi.org/10.1197/jamia.M1749
- Kapoor M. C. (2016). Types of studies and research design. Indian journal of anaesthesia, 60(9), 626–630. https://doi.org/10.4103/0019-5049.190616
- Ferguson, R. J., Terhorst, L., Gibbons, B., Posluszny, D. M., Chang, H., Bovbjerg, D. H., & McDonald, B. C. (2023). Using Single-Case Experimental Design and Patient-Reported Outcome Measures to Evaluate the Treatment of Cancer-Related Cognitive Impairment in Clinical Practice. Cancers, 15(18), 4643. https://doi.org/10.3390/cancers
- Bierer, S. B., Beck Dallaghan, G., Borges, N. J., Brondfield, S., Fung, C. C., Huggett, K. N., Teal, C. R., Thammasitboon, S., & Colbert, C. Y. (2025). Moving Beyond Simplistic Research Design in Health Professions Education: What a One-Group Pretest-Posttest Design Will Not Prove. MedEdPORTAL : the journal of teaching and learning resources, 21, 11527. https://doi.org/10.15766/mep_2374-8265.11527
- Chen, Z., Chen, K., Li, Y., Geng, D., Li, X., Liang, X., Lu, H., Ding, S., Xiao, Z., Ma, X., Zheng, L., Ding, D., Zhao, Q., Yang, L., & Alzheimer’s Disease Neuroimaging Initiative (2024). Structural, static, and dynamic functional MRI predictors for conversion from mild cognitive impairment to Alzheimer’s disease: Inter-cohort validation of Shanghai Memory Study and ADNI. Human brain mapping, 45(1), e26529. https://doi.org/10.1002/hbm.26529
- Chew B. H. (2019). Planning and Conducting Clinical Research: The Whole Process. Cureus, 11(2), e4112. https://doi.org/10.7759/cureus.4112
- O’Connell, N. S., Dai, L., Jiang, Y., Speiser, J. L., Ward, R., Wei, W., Carroll, R., & Gebregziabher, M. (2017). Methods for Analysis of Pre-Post Data in Clinical Research: A Comparison of Five Common Methods. Journal of biometrics & biostatistics, 8(1), 1–8. https://doi.org/10.4172/2155-6180.1000334
- Williams, B. (2024, December 27). Limitations of experimental research design – Insight7 – call analytics & AI coaching for customer teams. Insight7 – Call Analytics & AI Coaching for Customer Teams; Insight7. https://insight7.io/limitations-of-experimental-research-design/

