- Services
- Discovery & Intelligence Services
- Publication Support Services
- Sample Work

Publication Support Service
- Editing & Translation
-
Editing and Translation Services
- Sample Work

Editing and Translation Service
-
- Research Services
- Sample Work

Research Services
- Physician Writing
- Sample Work

Physician Writing Service
- Statistical Analyses
- Sample Work

Statistical Analyses
- Data Collection
- AI and ML Services
- Medical Writing
- Sample Work

Medical Writing
- Research Impact
- Sample Work

Research Impact
- Medical & Scientific Communication
- Medico Legal Services
- Educational Content
- Academic Editorial Services
- Educational Editorial Service
-
Education Editorial Services
-
- Industries
- Subjects
- About Us
- Academy
- Insights
- Contact Us
Biotechnology
At Pubrica, we provide end-to-end solutions for biotechnology research, development, and commercialization. Our expert teams help biotech companies navigate regulatory landscapes, optimize clinical strategies, and accelerate innovation with precision and compliance.
Empowering Biotechnology Innovation Through Expert Scientific and Regulatory Support
The biotechnology industry is at the forefront of scientific innovation, combining biology, technology, and data science to develop groundbreaking solutions for healthcare, agriculture, industrial processes, and environmental sustainability. From genomics and molecular diagnostics to regenerative medicine and biopharmaceuticals, biotechnology drives the development of therapies, vaccines, and technologies that improve lives globally.
With rapid scientific advancements, stringent regulatory requirements, and complex clinical pipelines, biotechnology organizations require expert guidance to navigate research, regulatory compliance, and commercialization. Pubrica specializes in supporting biotechnology companies across all stages of development, ensuring scientific integrity, regulatory compliance, and operational excellence.
Currently, there are more than 250 biotechnology healthcare products and vaccines available to patients, many of which treat previously untreatable diseases. More than 13.3 million farmers around the world use agricultural biotechnology to increase yields, prevent damage from insects and pests, and reduce farming’s impact on the environment.

Our Expertise in Biotechnology Includes
We provide end-to-end support for biotechnology companies, helping them accelerate research, ensure regulatory compliance, and bring breakthrough products to market efficiently. Our expertise spans multiple domains:
Biopharmaceutical Development
Guidance and support in biologics, biosimilars, vaccines, and therapeutic proteins, ensuring compliance with global regulatory standards.
Molecular and Cellular Research
Assistance with genetic engineering, cell therapy, gene editing, and molecular diagnostics, from preclinical studies to clinical translation.
Regulatory Affairs & Compliance
Comprehensive regulatory strategy, submissions, and documentation for biotechnology products across multiple markets.
Clinical Research & Trials
End-to-end clinical trial design, protocol development, and study management tailored to biotech therapeutics.
Scientific Communication & Publication Support
High-quality scientific writing, manuscript development, and regulatory communication tailored to complex biotech innovations.
Market Access & Commercialization Support
Strategic guidance for market entry, health economics, and reimbursement planning for biotech products.

Types of Biotechnology We Offer
Biotechnology is a broad field that applies biological systems, organisms, or derivatives to develop products and technologies for various industries. It can be classified into several types based on applications, techniques, and the focus area. Here’s a clear breakdown:
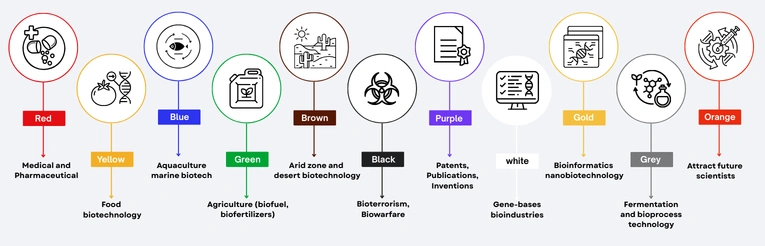
Biotechnology Segments We Serve
At Pubrica, we provide specialized scientific, regulatory, and publishing support across diverse segments of the biotechnology industry. Our services are tailored to meet the unique needs of each segment, enabling innovation, compliance, and high-quality dissemination of research. Key segments we serve include:




Why Pubrica for Biotechnology
 End-to-End Solutions
End-to-End Solutions
Support throughout the product lifecycle, from discovery to market access.
 Global Expertise
Global Expertise
Experience with submissions and compliance across multiple regulatory authorities worldwide.
 Tailored Approach
Tailored Approach
Customized solutions aligned with your product, therapeutic area, and development stage.
 Scientific Excellence
Scientific Excellence
Team of experienced scientists, medical writers, and regulatory specialists committed to quality and innovation.
Where Our Authors Publish
Our authors share Pubrica’s expert content in top-tier journals, conferences, and platforms, maximizing and amplifying its recognition and reach. Our placement will enhance our visibility and elevate our standing in an authoritative capacity.

Paper Title: Novel technologies and their applications in biotechnology and the life sciences.
Author: Deeni Y, Beccari T, Dundar M
Journal Name: Journal of Biotechnology
Publisher: Elsevier
Impact factor: 3.9
Our Expert Biotechnology Editors
Pubrica’s team of industry specialists offers unrivalled expertise and perspectives to provide complete solutions with precision and originality. Through a mix of both experience and specialization, they strive for excellence in everything they do.






What Our Client Says About Us
Pubrica’s deep understanding of the biotech sector allowed us to enhance our manuscript quality and compliance with international standards. Their support made a significant difference in our publication success.

USA

Working with Pubrica has been transformative for our biotech research team. Their expertise in regulatory documentation and scientific communication helped us streamline submissions and accelerate our projects.

India

The team at Pubrica brings exceptional precision and scientific rigor to every project. Their insights into biotechnology trends and regulatory requirements were invaluable to our R&D process.

Singapore

Testimonials
Learn how Pubrica’s meta-analysis service has empowered researchers to generate high-impact, publication-ready analyses that advance evidence-based research and elevate their academic and clinical visibility. Here is what our clients say:

"Pubrica’s team provided exceptional support throughout my meta-analysis cardiovascular drug efficacy. Their adherence to PRISMA guidelines and attention to statistical detail helped me publish in the European Heart Journal. Highly recommended"
— Dr. Anna Müller
Cardiologist, University Hospital Munich, Germany

"The meta-analysis manuscript I co-authored with Pubrica’s experts was accepted by BMC Public Health without major revisions. Their data synthesis and transparent methodology were critical to this success."
— Dr. Rohan Mehta
Public Health Researcher,
All India Institute of Medical Sciences (AIIMS), India

"Thanks to Pubrica’s guidance, our meta-analysis on paediatric nutrition was published in The Lancet Child & Adolescent Health. The methodological rigor and rewriting support were key contributors to the paper’s clarity and impact."
— Dr. Luis Fernández
Pediatrician & Research Fellow, University of Barcelona, Spain
Insights
How to Structure Case Reports and Review Articles for Medical Journals
Medical journals expect a structure for case reports and review articles, with clear objectives....
How Should Physicians Choose the Right Journal for Submitting a Case...
Publishing a case report involves more than clinical knowledge; it also demands strategic journal ....
How Physicians Can Write Clear and Impactful Patient Education Materials
Effective patient education materials (PEMs) are crucial for promoting health literacy, enhancing....




