- Services
- Discovery & Intelligence Services
- Publication Support Services
- Sample Work

Publication Support Service
- Editing & Translation
-
Editing and Translation Services
- Sample Work

Editing and Translation Service
-
- Research Services
- Sample Work

Research Services
- Physician Writing
- Sample Work

Physician Writing Service
- Statistical Analyses
- Sample Work

Statistical Analyses
- Data Collection
- AI and ML Services
- Medical Writing
- Sample Work

Medical Writing
- Research Impact
- Sample Work

Research Impact
- Medical & Scientific Communication
- Medico Legal Services
- Educational Content
- Academic Editorial Services
- Educational Editorial Service
-
Education Editorial Services
-
- Industries
- Subjects
- About Us
- Academy
- Insights
- Contact Us
How to Use the GRADE Framework in Evidence-Based Medicine: A Practical Guide
- Home
- Academy
- Research Services
- How to Use the GRADE Framework in Evidence-Based Medicine: A Practical Guide
High-Impact Journals
Interesting topics
How to Use the GRADE Framework in Evidence-Based Medicine: A Practical Guide
1. Understanding the Foundations of GRADE
2. The Five Domains for Rating Down Certainty
3. Rating Up Observational Evidence
4. Determining Overall Certainty of Evidence
5. From Evidence to Recommendations
6. Summary of Findings Tables and Transparency
7. Practical Workflow for Implementing GRADE
The GRADE framework (Grading of Recommendations Assessment, Development, and Evaluation) is a transparent, structured system used globally in healthcare to rate the quality (certainty) of evidence and determine the strength of clinical recommendations. It classifies evidence as high, moderate, low, or very low based on risk of bias, consistency, and precision, and categorizes recommendations as strong or conditional. The GRADE framework evidence-based medicine approach plays a central role in strengthening transparency and reproducibility in modern healthcare recommendations.
Evidence-Based Medicine uses evidential sources in such a way that they are verifiable and replicable. The GRADE framework developed by the GRADE Working Group is currently the most relied upon by healthcare providers in their clinical and policy decision making. GRADE provides an objective means through which health professionals can evaluate their level of certainty regarding various interventions’ effectiveness. This guide provides a structured method of assessing the level of confidence in an evidence-based decision-making process based on the use of GRADE. This structured approach is widely adopted in clinical practice guidelines development to ensure consistency and methodological rigor.
1. Understanding the Foundations of GRADE
GRADE distinguishes between two core judgments:
- Certainty (quality) of evidence
- Strength of recommendation
Unlike earlier hierarchies that relied mainly on study design, GRADE evaluates multiple domains that influence confidence in an estimate of effect.[1] Randomised controlled trials (RCTs) begin as high-certainty evidence, while observational studies begin as low-certainty evidence. However, ratings can change depending on methodological strengths or weaknesses. [2]
Key Principles
- Evidence is assessed per outcome, not per study.
- Certainty reflects confidence that the true effect lies close to the estimated effect.
- Recommendations consider more than evidence certainty alone.
The clinical practice guidelines GRADE method ensures that recommendations are not based solely on study design but on a comprehensive evaluation of evidence domains.
2. The Five Domains for Rating Down Certainty
GRADE evaluates five domains that may reduce confidence in evidence.
- Risk of Bias – Assesses the internal validity of the studies included in the systematic review and helps identify any limitations, such as randomisation being inadequate, blinding not being done, or selective reporting, which lead to a decrease in rating.[3]
- Inconsistency- If there is large heterogeneity (e.g., high I²) that cannot be attributed to sampling error, it decreases certainty in the evidence.
- Indirectness- Indirectness is when either the population, intervention, control, or outcome listed in your clinical question does not match one that is included in your systematic review.
- Imprecision- If the confidence intervals around the point estimate are wide and cross clinically important thresholds. [4]
- Publication Bias- Causes a distortion in the evidence base by only publishing studies that report positive results.
Example: Applying Downgrading
Clinical Question: Does Drug X reduce cardiovascular mortality?
- RCT evidence available → Start at High
- Serious risk of bias → −1 level
- Wide confidence interval → −1 level
Final Certainty: Low
This structured downgrading ensures transparency and reproducibility.
Assessment of Cochrane review evidence quality often incorporates these same domains to ensure methodological transparency.
3. Rating Up Observational Evidence
While observational studies begin at low certainty, they may be upgraded when:
- There is a large magnitude of effect
- A dose–response gradient is present
- All plausible confounding would reduce the observed effect. [5]
This flexibility allows high-quality real-world evidence to meaningfully inform clinical decisions.
4. Determining Overall Certainty of Evidence
GRADE classifies certainty into four levels:
Certainty Level | Interpretation |
High | Very confident in effect estimate |
Moderate | Likely close to true effect |
Low | Limited confidence |
Very Low | True effect likely substantially different |
The lowest rating across critical domains determines the final certainty for each outcome (Scheunemann et al., 2013). Importantly, certainty is assessed separately for each critical outcome (e.g., mortality, adverse events, quality of life). The PICO framework in evidence grading further strengthens clarity by aligning outcomes directly with clinical questions.
5. From Evidence to Recommendations
Building upon evidence to create recommendations involves systematic judgment that transcends statistical results. The GRADE Evidence-to-Decision (EtD) framework provides for the four factors to consider in its decision-making process:
- Balance between benefits and harms
- Certainty of evidence
- Patient values and preferences
- Economic implications of resources used
- Equity, feasibility and acceptability. [6]
Types of Recommendations
- Strong Recommendation – Most patients should receive the intervention.
- Conditional Recommendation – Shared decision-making is appropriate.
Notably, strong recommendations can occasionally arise from moderate or even low-certainty evidence when benefits clearly outweigh harms in critical contexts.[7]
Example: Evidence-to-Decision in Practice
Intervention: Oral anticoagulants for atrial fibrillation
- Benefit: Stroke reduction
- Harm: Increased bleeding risk
- Certainty: Moderate
- Patient values: Stroke prevention prioritized
Recommendation: Strong in favor
Integration with a clinical decision support system EBM enhances the translation of these recommendations into real-world healthcare settings.
6. Summary of Findings, Tables, and Transparency
GRADE clarifies the results by providing Summary of Findings (SoF) tables, which contain:
- The relative and absolute effect sizes
- Number of participants and studies
- Level of certainty (certainty rating)
- Explanatory footnotes describing the rationale for downgrading/upgrading decisions
SoF tables help clinicians and policymakers interpret results and are an integral part of systematic reviews and developing guidelines. Both the World Health Organisation and the Cochrane Collaboration support GRADE as a means of ensuring methodological rigor and global consistency across research.
7. Practical Workflow for Implementing GRADE
Below is a streamlined stepwise approach for applying GRADE in clinical research or guideline development:
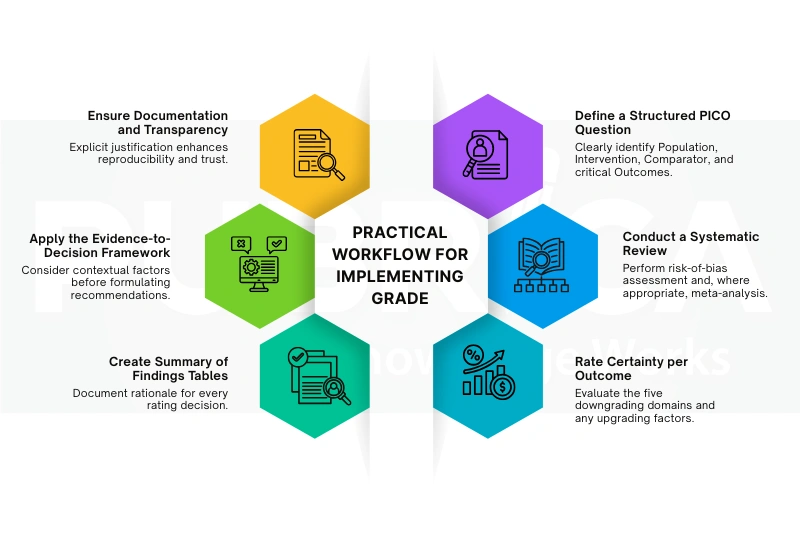
Modern clinical guideline development software can streamline this workflow by integrating grading, documentation, and reporting processes.
Connect with us to explore how we can support you in maintaining academic integrity and enhancing the visibility of your research across the world!
Conclusion
The GRADE framework offers an updated methodology in Evidence-Based Medicine by providing an accepted method for evaluating both the certainty and strength of health recommendations. The separation of evidence quality from the strength of recommendations; combined with the inclusion of various contextual factors, increases transparency and the applicability of clinical practice and reliability of policy. When implemented consistently, GRADE enhances credibility of guidelines, while also supporting global health care decision-making from an independent perspective.
Strengthen your systematic reviews and clinical guidelines with expert GRADE methodology support. Pubrica evidence-based research specialists help you develop high-quality reviews, Summary of Findings tables, and transparent recommendations aligned with global standards. [Get Expert Publishing Support] or [Schedule a Free Consultation].
References
- Guyatt, G. H., Oxman, A. D., Vist, G. E., Kunz, R., Falck-Ytter, Y., Alonso-Coello, P., Schünemann, H. J., & GRADE Working Group (2008). GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ (Clinical research ed.), 336(7650), 924–926. https://doi.org/10.1136/bmj.39489.470347.AD
- Guyatt, G., Oxman, A. D., Akl, E. A., Kunz, R., Vist, G., Brozek, J., Norris, S., Falck-Ytter, Y., Glasziou, P., DeBeer, H., Jaeschke, R., Rind, D., Meerpohl, J., Dahm, P., & Schünemann, H. J. (2011). GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. Journal of clinical epidemiology, 64(4), 383–394. https://doi.org/10.1016/j.jclinepi.2010.04.026
- Guyatt, G. H., Oxman, A. D., Montori, V., Vist, G., Kunz, R., Brozek, J., Alonso-Coello, P., Djulbegovic, B., Atkins, D., Falck-Ytter, Y., Williams, J. W., Jr, Meerpohl, J., Norris, S. L., Akl, E. A., & Schünemann, H. J. (2011). GRADE guidelines: 5. Rating the quality of evidence–publication bias. Journal of clinical epidemiology, 64(12), 1277–1282. https://doi.org/10.1016/j.jclinepi.2011.01.011
- Guyatt, G. H., Oxman, A. D., Kunz, R., Brozek, J., Alonso-Coello, P., Rind, D., Devereaux, P. J., Montori, V. M., Freyschuss, B., Vist, G., Jaeschke, R., Williams, J. W., Jr, Murad, M. H., Sinclair, D., Falck-Ytter, Y., Meerpohl, J., Whittington, C., Thorlund, K., Andrews, J., & Schünemann, H. J. (2011). GRADE guidelines 6. Rating the quality of evidence–imprecision. Journal of clinical epidemiology, 64(12), 1283–1293. https://doi.org/10.1016/j.jclinepi.2011.01.012
- Guyatt, G. H., Oxman, A. D., Kunz, R., Woodcock, J., Brozek, J., Helfand, M., Alonso-Coello, P., Glasziou, P., Jaeschke, R., Akl, E. A., Norris, S., Vist, G., Dahm, P., Shukla, V. K., Higgins, J., Falck-Ytter, Y., Schünemann, H. J., & GRADE Working Group (2011). GRADE guidelines: 7. Rating the quality of evidence–inconsistency. Journal of clinical epidemiology, 64(12), 1294–1302. https://doi.org/10.1016/j.jclinepi.2011.03.017
- Alonso-Coello, P., Schünemann, H. J., Moberg, J., Brignardello-Petersen, R., Akl, E. A., Davoli, M., Treweek, S., Mustafa, R. A., Rada, G., Rosenbaum, S., Morelli, A., Guyatt, G. H., Oxman, A. D., & GRADE Working Group (2016). GRADE Evidence to Decision (EtD) frameworks: a systematic and transparent approach to making well informed healthcare choices. 1: Introduction. BMJ (Clinical research ed.), 353, i2016. https://doi.org/10.1136/bmj.i2016
- Andrews, J. C., Schünemann, H. J., Oxman, A. D., Pottie, K., Meerpohl, J. J., Coello, P. A., Rind, D., Montori, V. M., Brito, J. P., Norris, S., Elbarbary, M., Post, P., Nasser, M., Shukla, V., Jaeschke, R., Brozek, J., Djulbegovic, B., & Guyatt, G. (2013). GRADE guidelines: 15. Going from evidence to recommendation-determinants of a recommendation’s direction and strength. Journal of clinical epidemiology, 66(7), 726–735. https://doi.org/10.1016/j.jclinepi.2013.02.003

