
Pre-or post-publication Peer review of Scientific Manuscripts: Thoughts on Pros and Cons
October 31, 2019
Guidelines to authors in Writing an effective Medical Case Report (MCR)
November 6, 2019Structure for writing an effective clinical review articles
Clinicians need clinical review articles that have well-organized structure for easy reading and understanding. Only then it will help them to apply it practically on patients and for further research. Traditional clinical review articles, also known as updates, discuss a topic broadly and selectively review the medical literature. An excellent structured clinical research article will resolve controversy generated by studies that contradict one another.
Before knowing how to structure a clinical review article, you have to know some background facts on why good structure is crucial for it.
For clinicians to upgrade their knowledge, they rely only on peer-reviewed medical journals. But most studies at best only provide preliminary evidence due to
- Limited scope
- Poor design
- Inefficient execution
- Inadequate sample size to have
significant clinical benefits or detection of adverse effects - Play of chances
Clinicians reading these studies, have to integrate and compare it with existing evidence to conclude, whether the clinical policy had to be changed on the accumulated evidence. Even in the case of original studies, only a portion of the clinical problem is addressed. Hence, most clinicians take a short cut to read the clinical reviews done by others. These clinic review articles include evidence from available studies on the specific clinical problem.
But the clinical review if not structured properly, will not do justice to the original evidence. And the clinician will end up with false conclusions which will finally be borne by the patients. In research by Mulrow conducted on 50 published reviews, only one adhered to the specific methods of identifying, selecting, and validating included information.
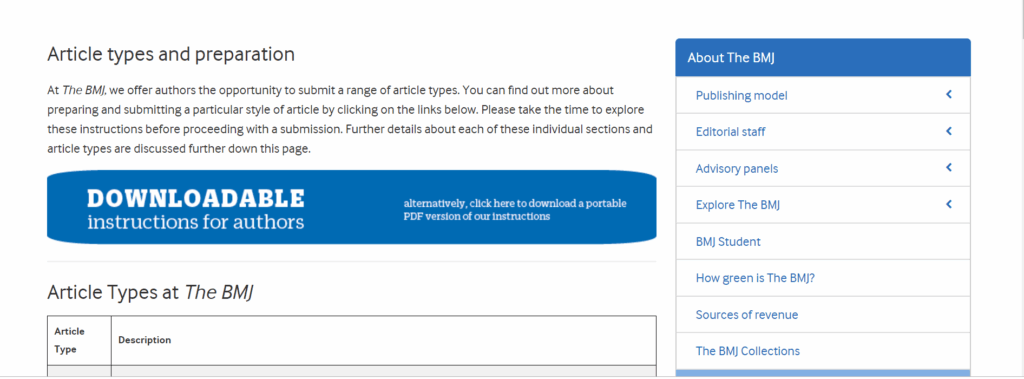
| Article Type | Description | Abstract Structure | Abstract Word count | Article Structure | Article Word count |
| Full length Articles | Original research and/or clinical studies. Please note, as described in the instructions for authors, papers with only present data specific to a particular geographical area or country which are not novel or surprising from a worldwide perspective, are unlikely to be published. | –Background -objectives -study design -Results -Conclusions | 250 | –Background -objectives -study design -Results -Discussion (Acknowledgement)COI | 2500 |
| Short communication | Original research and/or clinical studies that do not require a full paper, but are completed studies, may be submitted as Short Communications. These papers may detail a smaller number of observations or may include a smaller number of patients. They may also add a small amount of new information but which is still considered important. | -Background -Objectives -Study design –Results -Conclusions | 250 | -Background -Objectives -Study design -Results -Discussion (acknowledgements) COI | 1250 |
| Case Reports | Short articles of a clinical nature which illuminate an underlying principle of a disease state, its diagnosis, or its therapy | -Why this case is important -Case description -Other similar and contrasting cases in the literature -Discussion and references | 1500 | ||
| My Favourite Assay | This offers an opportunity for researchers to present an assay that they have developed and that they feel would be useful for others | -Reasons for developing the assay -Methods used in designing the assay -Protocol -Validation data | 500 | ||
| Letters to the Editor | Comments on previously published articles or any other items of interest to clinical virologists | 500 | |||
| Guidelines and clinical algorithms | Documents that have been locally or internationally approved and contain general principles that may benefit others in their clinical practice or in writing their own algorithms or guidelines | No limited | |||
| Commentaries and points of view | Please contact one of the Editors -in -Chief to check on the suitability of your topic | 750 | |||
| Review articles | Reviews should give a succinct overview of a particular topic. Please contact one of the Editors in-Chief to check on the suitability of your topic | Unstructured abstract | 250 | 3000 | |
| Meeting reports, News &Announcements: – Meeting reports | Summary of a relevant symposium, workshop, etc. | 1500 | |||
| VIROQAS | This should be a clinical case that presents a diagnostic dilemma that will impart an important learning point | – Part I: Case presentation. Questions for readers at end (bold) – Part II: Evidence-based opinion. Each subsection starts with question from Part I – Page break between Part I and II | 1250 |
Notes: – The word count in the Article structure excludes the Abstract, References, Acknowledgement and figure captions. The word count should be indicated at the foot of the title page. – Keywords (3-6) should be provided at the foot of the abstract.
Now let us see how a clinical research article can be structured:
PRISMA or Preferred Reporting Items for Systematic Reviews and Meta-analyses has issued a statement listing 27 item checklists to make the writing of clinic review feasible.
Though IMRAD (Introduction, Methods, Results, Discussion and Conclusions) structure is the basis of many reviews, there could be variations in guidelines as per the nature of research. But the general structure for most of the clinic review structure is as follows
Elements of a review article
1. Title page:
- Length: The title should be in a range of 8 -12 words
describing the topic and its aspect.
E.g. The title “Pancreatic Disease” will be too generic. Instead, “Challenges in Diagnosing Subclinical Pancreatic Disease” will describe the concise nature and aspect of the research.
- The title must be short
- The title should have all
the critical elements of the subject matter (or informative) - List of authors: Declare intellectual ownership of the work, provide contact
information: Author name &affiliations- Order of authors: The first
author has done most of the research - Second and in between authors
would have contributed one way or another - The last author coordinated the
project
- Order of authors: The first
- Tense: Results: the present tense stresses the general
validity and demonstrates that the author is trying to achieve with the
article. The paste tense indicates that results are not
established yet. - Citations: None
2. Abstract:
- Citation: No citations needed in the abstract
- Elements: Descriptive Abstract for narrative reviews
- Tense: Present
- Objectives: Present
- Materials and Methods, results:
Past - Conclusion: Present
- Should include acronyms and
abbreviations only if they are used more than once - Length: It should be written within a range of 200 – 500 words outlining
the main points or synthesis in subheadings as per the nature of the review
E.g., Abstract requirements for content and format differ according to the type of review or journal. Some prefer unstructured abstracts, and the others require structured abstracts containing the elements in subheadings.
- Journal of Clinical
chemistry reviews require a structured abstract with subheadings
background, content, and summary- Another style needs the
subheadings content, evidence, acquisition, results, and conclusions
- Another style needs the
3. Introduction:
- Tense: Should be written in present tense addressing the research question
and the objective purpose of the review - Citations: Many
- Length: 10% and 20% of the core text
- Note: It should explain why the
review of the field or topic essential at this time and what is that you are
going to cover in the review
E.g., Review on DMS or deep brain stimulation for Dystonia
However, one must acknowledge that published results obtained with DBS in Dystonia are few and that conclusions from these preliminary reports should be drawn very cautiously. Nonetheless, promising findings are emerging from single case reports or small case series, and the notion that DBS may be of great help in selected cases is progressively growing… . In this review, we discuss the results reported in the literature. Some critical issues regarding the evaluation of the results are also mentioned
4. Materials and Methods
Tense: Should be written in the past tense to provide the reason to repeat the review
Elements: It should include:
- Search strategies
- Criteria of inclusion and
exclusion of methods - Data sources
- Geographical information
- Characteristics of study
subjects - Details of used statistical
analyses
Length: Approx. 5% of the core text
Citations: Few (e.g. software and statistical analyses used)
5. Main Body Part of the Review Article
Section Structure: A coherent structure of the topic mentioning their relevance to the objective. Subheadings reflecting the topic organisation and indicate the content in various sections including methodological approaches adopted, theories or models used, critical appraisal of studies, in chronological order
Links: Links the research findings to the research questions
Tense: Three tenses are frequently used: Present tense, when reporting what another author thinks, and writes. Example: It is believed….
Present Perfect, when referring to an area of research with a number of researchers Example: They have found….
while simple past referring to what a specific researcher found, referring to a single study. Example: They found….
Citations: Usually indirect but can also be cited directly
Length: 70 to 90% of the core text
6. Discussion:
Elements: Discuss the results and their significance clearly and concisely. Identify any unresolved questions.
Function: Reiterate the objective and background information
Citations: Few or more
Tense: Present tense
E.g.
Despite 30 years of continued investigation, the precise mechanism of CD4 T-cell loss induced by HIV infection remains controversial. HIV-mediated destruction of its preferred target, the activated CD4 T cell, is certainly central to HIV pathogenesis, but does not explain why many uninfected cells die or why the host cannot merely replace lost cells.21,22 As first proposed in the 1990s,23 researchers now know that the pro-inflammatory nature of HIV infection is a key part of disease pathogenesis.2,25
7. Conclusions:
- Conclude the review to give the
reader a sense of what the review means by discussing the objective mentioned
in introduction, including findings and interpretations. - I should also discuss what the
future is by identifying the unresolved questions
E.g.
Future studies evaluating novel stroke biomarkers should answer questions that address their unique clinical contribution in the diagnosis, management, and risk prediction of stroke: has the patient had a stroke? Is the stroke of ischemic or hemorrhagic etiology? Are symptoms suggestive of additional intensive investigation or thrombolytic therapy? Is the patient at risk for stroke or reoccurrence of cardiovascular events? Modern stroke diagnosis remains heavily reliant on clinical interpretation, and further translational research efforts toward discovery of stroke biomarkers have the possibility to greatly improve patient outcomes and quality of care
8. References:
Length: References should be written in 50 to 100 words and could cite only those in text, and online sources are not allowed.
Plagiarism: Strictly Avoid
The above details will help you to structure your clinical review article and if you need any further assistance seek professional help.
Tags:
clinical review articles | medical literature | clinical case | Schemantic reviews | Meta analysis | writing of clinical review | clinical review structure







