- Services
- Discovery & Intelligence Services
- Publication Support Services
- Sample Work

Publication Support Service
- Editing & Translation
-
Editing and Translation Services
- Sample Work

Editing and Translation Service
-
- Research Services
- Sample Work

Research Services
- Physician Writing
- Sample Work

Physician Writing Service
- Statistical Analyses
- Sample Work

Statistical Analyses
- Data Collection
- AI and ML Services
- Medical Writing
- Sample Work

Medical Writing
- Research Impact
- Sample Work

Research Impact
- Medical & Scientific Communication
- Medico Legal Services
- Educational Content
- Academic Editorial Services
- Educational Editorial Service
-
Education Editorial Services
-
- Industries
- Subjects
- About Us
- Academy
- Insights
- Contact Us
Targeted literature searches are a fundamental part of writing clinical manuscripts that will meet the standards of high-quality journals and contribute meaningfully to evidence-based practice. When physicians write clinical manuscripts, utilizing a targeted literature search can identify high-quality, relevant, and current evidence. While a general literature review is useful, a targeted literature search is specific to the clinical question and should be completed through frameworks established, such as PICO (Population, Intervention, Comparator, Outcome) and PRISMA [1].
Navigating the Complexities of the Pharmaceutical Industry: Drug Development, Pricing, and Marketing
- Home
- Academy
- Research Impact
- Navigating the Complexities of the Pharmaceutical Industry: Drug Development, Pricing, and Marketing
High-Impact Journals
Interesting topics
Navigating the Complexities of the Pharmaceutical Industry: Drug Development, Pricing, and Marketing
The pharmaceutical industry functions in a complicated environment in which drug development, pricing decisions, and marketing strategies can be affected by several factors. All stakeholders must understand how these factors work together to navigate the complexities of the healthcare landscape.[1]
1. Drug Development Process
- Discovery and Preclinical Research: This is the initial stage of the timeline for identifying possible drug candidates and laboratory research into the efficacy and safety of drug candidates.[2]
- Clinical Trials: Drug candidates undergo testing in humans in Phases I-III, where their safety, dosage, efficacy, and possible side effects are evaluated.[3]
- Regulatory Review and Approval: Once trials are complete, the data is submitted to regulatory authorities like the FDA for approval of the drug before marketing.
- Post-marketing Surveillance: Drugs are monitored for overall and extended effects in the general population even after approval.[4]
2. Drug Discovery and Development Process
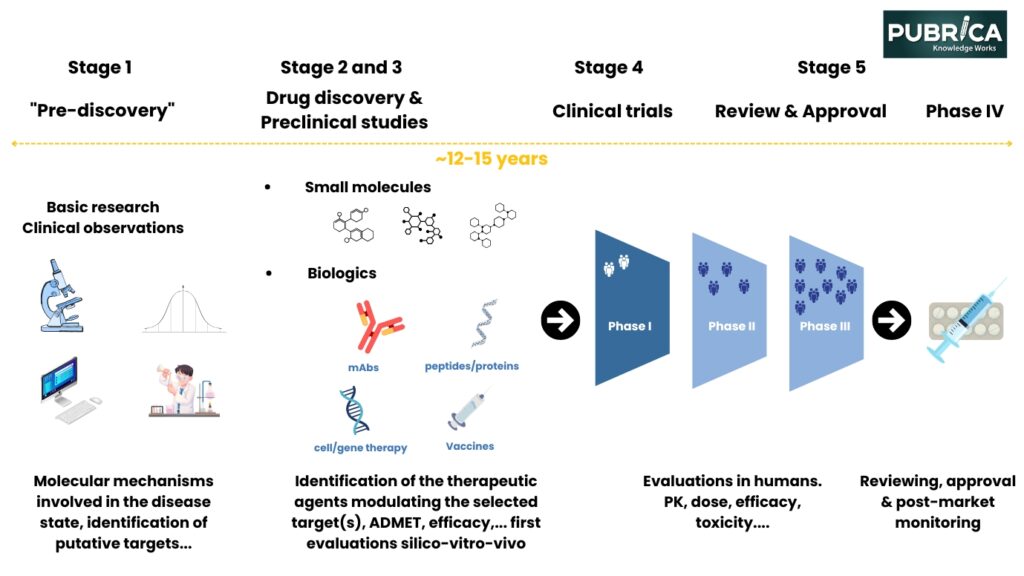
Image adapted from Singh N, Vayer P, Tanwar S, Poyet JL, Tsaioun K, Villoutreix BO. Drug discovery and development: introduction to the general public and patient groups. Front Drug Discov (Lausanne). 2023;3(1201419). doi:10.3389/fddsv.2023.1201419
3. Factors Influencing Drug
Pricing Decisions
| Research and Development Investments | The cost of designing a new drug is often substantial, often amounting to billions of dollars. Research and development costs are key factors in determining price. |
| Market Alternatives | The presence of market alternatives can cost, while the absence of a competitive market allows for a higher price. |
| Regulations | The government will examine your price, either through price controls or reimbursement rates. These regulations can be the key driver for price. |
| Health Technology Assessments (HTA) | Institutions such as the Institute for Clinical and Economic Review (ICER) can examine the cost and benefit of drugs for Pharmacoeconomics. |
| Insurance and Reimbursement | The big three will look at willingness to pay (will insurance cover that drug), and what you can get for a particular offer. |
4. Marketing Strategies in the Pharmaceutical Industry
Pharmaceutical marketing strategies focus on building trust: [5]
- Direct-to-Consumer Advertising: Firms use a variety of media to advertise directly to patients to stimulate demand.
- Healthcare Professional Engagement: Pharmaceutical representatives provide education and information to healthcare professionals regarding new drugs to shape prescribing habits.
- Patient Assistance Programs: Companies create programs to enhance access, which assist patients in being able to pay for their medications, thereby indirectly impacting market reach.
- Digital Marketing: Companies use marketing strategies involving online platforms and social media to reach greater numbers, targeting specific audiences.
5. Regulatory Influence on Marketing
- Advertising Regulations: Agencies such as the FDA enforce regulations that provide guidelines for agent when they do advertising, with the condition that their ads must not be deceptive or misleading. In recent months, the FDA has ramped up its focus on pharmaceutical ads and has cracked down on unlawful advertising practices.
- Transparency Requirements: Drug companies or manufacturers must be transparent about adverse effects and other risks associated with a drug’s label. These regulations exist so that consumers have access to truthful and fair information.
- Social Media Regulations: Drug advertising regulations now extend to digital media as well, so any company that utilizes digital promotions must comply with the same regulations.
6. Case Study: GLP-1 Weight-Loss Drugs
GLP-1 Weight-Loss Drugs
- Drug Summary: GLP-1 receptor agonists such as semifluid (Ozempic, Wegovy) and tripeptide (Mounjaro) have shown efficacy in weight loss and cardiovascular benefits. [6]
- Pricing Trends: The Institute for Clinical and Economic Review (ICERh) has determined that these drugs are cost-effective, which will factor into pricing as they enter the market.
- Market Trends: Although costly, the medications are becoming popular, and there are questions about pricing and accessibility.
7. Ethical Considerations
- Access and Equity: High drug prices create access issues for some populations and raise worries about equity in healthcare.
- Transparency in Pricing: There is an increasing call for pharmaceutical companies to be open about the metrics for how drug prices are calculated.
- Influence on Prescribing: Marketing may impact healthcare providers who prescribe drugs by changing habits to use brand rather than generic.
Connect with us to explore how we can support you in maintaining academic integrity and enhancing the visibility of your research across the world!
Conclusion
The relationship between drug development, price decision-making, and marketing processes is multilayered and complex. Development, prices, and marketing are influenced by multiple parameters, including regulatory environment, market forces, and ethical considerations. Stakeholders in the drug development should recognize the intricacies of this environment, while at the same time being concerned with the need for drugs to be accessible, affordable, and marketed ethically.
Your Influence on Drug Development Pricing Decisions and Marketing? Our Pubrica consultants are here to guide you. [Get Expert Publishing Support] or [Schedule a Free Consultation]
References
- (N.d.). Nber.org. Retrieved September 16, 2025, from https://www.nber.org/reporter/2018number3/economics-drug-development-pricing-and-innovation-changing-market
- What is Preclinical Research and Why is it so Important?(n.d.). ALS Therapy Development Institute. Retrieved September 16, 2025, from https://www.als.net/news/what-is-preclinical-research/
- Sun, D., Gao, W., Hu, H., & Zhou, S. (2022). Why 90% of clinical drug development fails and how to improve it?. Acta pharmaceutica Sinica. B, 12(7), 3049–3062. https://doi.org/10.1016/j.apsb.2022.02.002
- Vlahović-Palčevski, V., & Mentzer, D. (2011). Postmarketing surveillance. Handbook of experimental pharmacology, 205, 339–351. https://doi.org/10.1007/978-3-642-20195-0_17
- Yuanjia, H., Ung, C. O. L., Ying, B., & Yitao, W. (2007). Marketing Strategy: The Chinese Pharmaceutical Market: Dynamics and a Proposed Investment Strategy. Journal of Medical Marketing, 7(1), 18-24. https://doi.org/10.1057/palgrave.jmm.5050061
- (N.d.). Fda.gov. Retrieved September 16, 2025, from https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/fdas-concerns-unapproved-glp-1-drugs-used-weight-loss

